Re-writing Nature’s Recipe Book: How Ginkgo Bioworks is Poised to Upset Almost Every Industry You Can Think Of
Ginkgo Bioworks, based in Boston, has been in the headlines lately:
- Ginkgo will “bring biodesign to the masses”. (Fast Company.)
- Ginkgo will “convince you to love GMOs”. (The Atlantic.)
- Ginkgo are the “DNA Cops” helping to “stop a new class of biological weapons from being created”. (Bloomberg.)
- Ginkgo will “reinvent Moore’s Law”. (Forbes.)
What does all this mean? How can one company do all that?
I recently spoke with Ginkgo Bioworks co-founders Tom Knight and Jason Kelly about the future of manufacturing and how the secrets of biology will be the secrets to unlocking the next Industrial Revolution.
Ginkgo Bioworks is More Than A Biology Company — They’re Assembling Instruction Manuals For the Entire Natural World
Tl:dr: Biology is nanotechnology that works.
Named after the Ginkgo tree, Ginkgo Bioworks is trying to bring industrial efficiency to biology and biological efficiency to industry by rethinking how almost everything is made.
Co-founder Tom Knight’s path from computer hardware to molecular biology is the stuff of legends, and is critical to understanding the Ginkgo story. Knight began a decades-long affiliation with MIT while he was still in high school, when he took some of the first ever computer programming classes and spent his summers in the then-nascent artificial intelligence lab. He ultimately graduated through an MIT PhD onto the MIT faculty, where he worked on the ARPANET — a very early version of the internet — and co-founded startups to commercialize his computer hardware inventions. One of the companies he co-founded — Symbolics — had the first registered .com domain name and eventually went public.
While teaching an integrated circuit design course, Knight challenged his students to predict the end of Moore’s Law — the operating principle that computing power would double just about every two years. Knight saw that the end of that pace of improvement was very near and wondered, ‘What’s next? What do you do about the fact that the core technology, which is the basis of high performance computing, is coming to an end?’”
He began to hunt for the answer, and found it in molecular biology. At 50 years old, with a PhD and decades of work at the highest levels of the computer engineering fields, he went back to school, starting with a sophomore level molecular biology class. He eventually founded a molecular biology lab within the computer science department at MIT, and began to test his hypothesis around leveraging biology to solve problems he’d previously contemplated using silicon.
They say necessity is the mother of invention, and Knight bumped up against a need: to run his experiments on gene-edited bacteria (more on that later,) he first needed to create the bacteria. It was time consuming, repetitive work, and his engineering brain produced an epiphany: there had to be an engineering solution to create gene-edited bacteria in a scalable, efficient way. At its core, his epiphany pointed to a critical question: what separates bio- and genetic engineering from physical engineering? What becomes possible if we think about them in the same way?
Knight’s decades of computing and engineering expertise applied to relatively new education in molecular biology led to a critical insight: applying an engineering mindset to the discovery process — making it possible to more rapidly and predictably engineer genomes — would take industrial microbiology to a new scale.
The Power of Bacteria
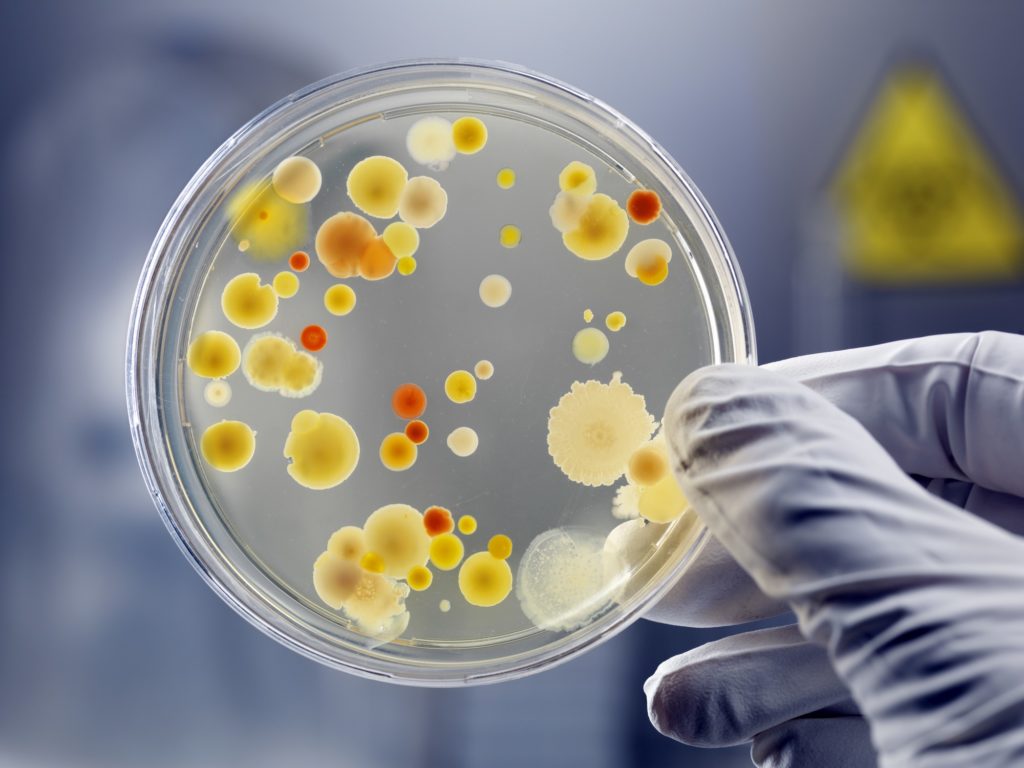
Ginkgo focuses on bacteria. It turns out that if you give these tiny prokaryotes the right instructions, they can make almost anything biology can.
“When we think about all the things we need in industry — energy storage, converting electrons into chemicals, etc — it turns out there are plenty of bacteria that have already solved these problems,” Knight told me.
Let’s start at the beginning: everything that is made in the biological world follows a specific recipe, like an instruction manual. Peptide A plus protein B plus phospholipid C plus nucleic acid D and voila, you’ve got an organism (like a plant), a protein (like keratin, the key structure that makes up our hair and the outer layer of our skin), or a chemical defense mechanism (like caffeine, which is a way plants make themselves unattractive to insects, but also happens to boost our cognition).
Building instruction manuals for bacteria — thousands of them, developed by rapidly engineering and prototyping generation after generation of gene-edited bacteria — is an efficient, cost-effective, and scalable process for making biological material. This is the motivating vision for Ginkgo. Tom, Jason, and their team want be a kind of Foxconn for biology, able to manufacture almost anything in the world cheaply, quickly, and at global scale.
Knight offered an eye-opening analogy: the utility of engineering biological molecules is “very similar in many respects to some of the basic motivations for technology like silicon, in the semiconductor world. There you have a base technology, in the case of silicon the ability to design and pattern and power-up semiconductor devices, very complex ones. The technology is agnostic to the application: you could be using it for making microprocessors or for cars. You can make television sets out of it. You can make iPhones.”
“One of the things we’re capitalizing on here at Ginkgo, is the generality of the platform,” Kelly added, “We’re designing biological molecules without a specific application in mind, knowing that we’ll use that technology across a whole variety of industries,” Kelly explained.
Ginkgo’s generalized platform will do for biology what the fabrication lab did for the semiconductor world: pave the way for automated technological production.
“In general, you can take the biology that is applicable to farm animals and apply it to plants. You can take the same technology that’s applicable to brewing beer and make antibodies with it,” Kelly explained, “because they all run on the same operating system — DNA.”
But what about all those headlines? The future of manufacturing? Biodesign? Bioweapons? DNA cops? All from bacteria?
That’s right, all from bacteria operating according to highly specialized instruction manuals, developed at scale according to a model unique to Ginkgo that applies a technological engineering approach to the task. More on the model in a bit, but first, Knight and Kelly took me through a few examples of what’s in process..
Example #1: A Little Bit Jurassic Park, A Little Bit Fern Gully, a Little Bit Chanel №5
Have you ever wondered what flowers smelled like in the Cretaceous? Would you buy Eau de Fern Crushed Under Your Feet During the Cambrian?
Using samples from the Harvard Herbarium, Ginkgo is hoping to produce novel scents for perfumers. By extracting DNA from flowers that have been extinct for hundreds of years, encoding that DNA in bacteria and growing it in yeast, they believe they can bring to market fragrances no living humans have ever smelled.
One major benefit of moving production into bacteria is speed. Which in industry is of course the same thing as cost. It takes months or even years to grow generations of plants, whereas bacterial generations can be produced in under 30 minutes.
Even if you’re not into Victorian-era scents, the ability to produce a scent via genetic manipulation of bacteria has important ramifications. Without it, if a perfumer wants to buy a fragrance molecule, they need to depend on farmers to harvest the whole plant. One ounce of Chanel Number 5 requires more than a thousand flowers. Extrapolate that across every product in every industry reliant on scents — from rose water to detergent to lotions, and yes, perfumes — and that’s a lot of crops. Relying on whole plants for scent molecules is wasteful and environmentally expensive, and, let’s face it, it sounds so 20th century, doesn’t it?

“Sometimes those crops don’t work for some reason during a particular year. And so if you’re a perfumer and you’re depending upon that crop, it can be a serious problem,” Knight said.
But if you take the fragrant chemical’s instruction manual, plop it into a bacteria, and print any bespoke chemical — ancient, modern, or maybe even brand new — on demand, you’ve just revolutionized a billion-dollar industry and removed the need for water and space-craving crops.
Example #2: We Can Make All Plants Into Self-fertilizing Plants
Let’s think bigger. Saving-our-planet bigger.
One of Ginkgo’s flagship projects, Joyn Bio, is a joint venture with Bayer to engineer a new class of nitrogen-fixing bacteria which would bind to the roots of corn and wheat and greatly lessen their need for fertilizer.
The idea is to transfer the microbial skills from one species (say, soybeans, which can trap their own nitrogen via bacteria that live in their roots) to another species of plant (wheat and corn, which can’t trap their own nitrogen), translating the instruction manual from one species to another.
If it works, corn and wheat crops will soon be able to fertilize themselves and replenish the soil, greatly reducing the amount of energy needed and greenhouse gasses emitted to sustain the world’s food supply.
Example #3: Biosecurity Meets National Security
As a side effect of being the largest designer of DNA in the world, Ginkgo has a large internal database of designed DNA, which gives them a library of sequences to compare any new unknown sequence in a cell, organism, virus, or pathogen. As part of a group in partnership with IARPA, Ginkgo is hoping to be able to answer questions about any new organism or DNA strand the government or health sector may come across, including whether it may be pathogenic (i.e. disease-causing), whether or not it was “man made” or whether or not it may be dangerous in some other way.
Example #4: A Fantastic Voyage Into the Body Could Revolutionize Medicine
The same idea — swapping instruction manuals — behind the production of custom or ancient fragrances and modified soil microbes could be applied to making medicines, too. Ginkgo, in a partnership with Synlogic, is exploring how gut bacteria can modulate disorders of both the body and mind. Like the Joyn Bio project is about engineering microbes that live with plants, this one is about engineering microbes that live in the gut. Ginkgo may one day be able to turn our gut bacteria into pharmaceutical factories — able to print any drug, cheaply, on demand, from inside the body.
“We might be able to do that by, again, copying what might be extracted out of a plant and moving that production capability into yeast or bacteria,” said Knight.

Image credit: Ginkgo Bioworks
The Ginkgo Difference
The possibilities are remarkable. So too is the Ginkgo model that converts potential into reality. It’s helpful to understand how they’re different than the typical biotech company.
The industry standard process goes like this: first, sign a contract to make a specific compound, for a specific purpose. Then, hire an army of molecular biologists and seat them at lab benches where they design and test genetic pathways. If you hired really good molecular biologists, they might be able to fairly quickly design, assemble, and test a half dozen pathways to find one or two that work for the specific purpose they’re hunting for.
Ginkgo turns that process on its head, thanks to Tom and Jason’s engineering mindset. Ginkgo works much more like in the silicon world — they treat the biology as an outcome-agnostic technology, rather than as a limited purpose-specific tactic.
So at any given time, Ginkgo teams are working on dozens of projects, producing pathways and organisms that could be applied to any number of specific purposes. Remember how silicon is agnostic to whether it’s powering an iphone or a car? Same here. The basic platform of bacteria, instructed in countless ways, is as agnostic to whether it’s powering medicine or scents or nitrogen-fixing roots? This is how Ginkgo has that vast library of DNA I described in example #3. This is the bacteria factory.
When Ginkgo gets a new project, they don’t need to hire a team of PhDs to sit at lab desks. Instead, they spin up an internal team to engage the factory to quickly design pathways, do the gene editing, apply it to generations of bacteria or yeast, and test and analyze them. If the designed organisms are producing the compound they’re looking for, they’re transitioned to a facility that produces them at scale. If not, they can rapidly repeat the process.
“So all along that path, there is a set of teams that are not project-specific but rather technology-specific in exactly the same way that you would find in the semiconductor fabrication facility where you have people who are good at designing masks and then people who are good at baking silicon wafers and then another set of people who are good at slicing those wafers up and packaging them,” Knight noted.
Kelly added, “Now notice that the team at each stage of production is good at what they are responsible for, and it doesn’t make any difference whether they’re making silicon for an automobile or a cell phone. The sequence of things that gets done is exactly the same, regardless of what the end product is.”
The Ginkgo team has made bioengineering “real engineering” — just as Tom hoped.
Ginkgo is Designing the Future
I asked Knight to think a little sci-fi. What will the future be like, with a Ginkgo Bioworks in charge?
Knight imagined growth in the field of predictive biology, where the designs can be even more efficient by simulating whether different instruction manuals will work, in which organisms, and when.
Outside of our own bodies printing medicines we need, self-fertilizing plants, or resurrecting fragrances, what might we imagine? Designer trees whose structural blueprints have been reprogrammed to grow into house shapes instead of log shapes? (I’m in. I want to live in a gigantic treehouse.) Roads made of self-healing concrete?? Almost all the world’s chemicals and energy being made in huge, environmentally-friendly, quiet, and clean silos in the middle of America?
As Knight noted, “Asking the question of what’s to come for synthetic biology is like asking Bardeen, the inventor of the transistor to ‘please predict iPhones’ in 1948. You can’t predict it. You can make guesses what’s important today but we don’t have a clue for the future.”
In other words, the sky… and the ground, and the seas, and humanity…essentially, the world, is the limit.
Share
Return to News